
Risking Your Future: Why Commercial Genetic Testing Is Disempowering
Genetic testing has been commercialised by companies such as 23andMe. They claim their services allow you to gain knowledge about your risk of getting a disease or negative lifestyle factors — for example, diabetes, Parkinson’s, sleep movement or genetic weight (23andMe, n.d.).
Risky society
The obsession with risk, as well as any intervention to decrease this risk, is a central topic in our society. This phenomenon has been coined risk society (Beck, 1992; Lupton, 2013). It is a neoliberal idea which gives people the responsibility to take care of their own health and assigns blame if they don’t.
The obsession with risk, as well as any intervention to decrease this risk, is a central topic in our society
Genetic testing companies jump onto the phenomenon of risk society and draw upon the idea that knowing risk empowers you to “live [your] healthiest life” (Teladoc Health, n.d.) or “own your health” (23andMe, n.d.). You become, as Harvey (2010) calls it, a genetic entrepreneur; a person who creates their own optimal healthy future by maximising the wellness specific to their own genetic makeup. The genetic entrepreneur visualises undesirable future states (e.g. I am likely to develop heart disease) and can avoid this from happening (e.g. I will stop smoking because of my increased risk). This claim makes four assumptions, which I will discuss in this article.
Assumption 1: It is possible to define diseases or lifestyle factors in terms of genetic risk
Note that commercial genetic testing is implemented in isolation. You provide DNA samples to a company, who gives you answers solely based on that DNA sample. Also note how only studying DNA is different from the clinical context, where genetic testing often is used along broader diagnostics — looking at family history, environmental factors, symptoms etc.
These two observations already mitigate a large part of the potential for commercial genetic testing. The claim that diseases or lifestyle factors are genetically determined rarely holds (Janssens et al., 2008). This is why most commercial genetic tests have limited clinical significance (Mathews et al., 2012; McGuire & Burke, 2011; Weedon et al., 2021).
The claim that diseases or lifestyle factors are genetically determined rarely holds
However, it would be unfair to claim the opposite (i.e. genetic tests have no clinical significance). It is true that some diseases are (partially) genetically determined — such as breast or ovarian cancer, or Parkinson’s disease. On top of this, we may be able to link more genes to diseases or lifestyle factors in the future. Saying genetic testing is ineffective, is an intuitive rebuttal but simply not sufficient to claim any harm.
Assumption 2: You are able to act upon risk information
In order for you to change your behaviour effectively it is necessary the information is both accurate and interpretable. However, genetic testing companies fail to comply with either.
False marketing
The United States Government Accountability Office (2010) found multiple examples of companies that market their tests as diagnostic tools or claim that their supplements are able to change DNA. Even in the case of cancer testing — which has clinical validity — genetic testing has been marketed to vulnerable groups with the use of anxiety-evoking strategies through misrepresentation and omission (Hull & Prasad, 2001). Even if these exact problems are solved, companies are still here to make a profit, rather than to provide accurate information.
Genetic testing has been marketed to vulnerable groups with the use of anxiety-evoking strategies through misrepresentation and omission
The Black Box Called Genetics
Assuming a full understanding of genetics by a normal consumer is rather ambitious — even though some companies do assume it (e.g. Genos).
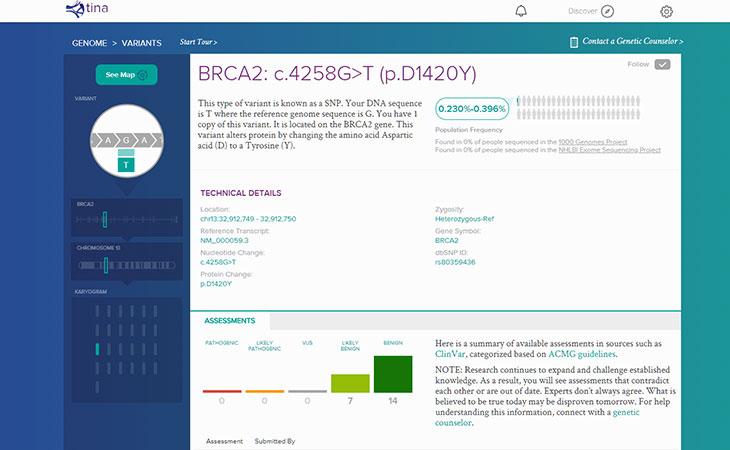
Genos' interface with data on each variant
Instead, companies often report a limited amount of information, such as information on relative risk without validity information (e.g. 23andMe). In both cases, you are left behind without the knowledge necessary to act.
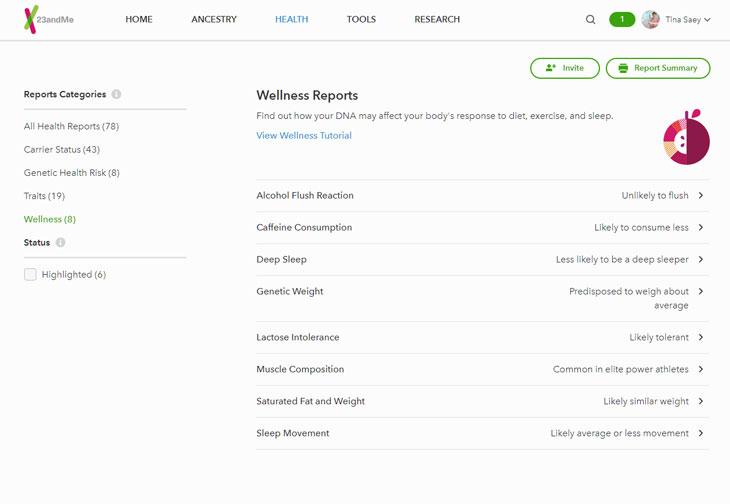
23andMe's interface with only relative risks
The lack of knowledge of genetics creates a need for genetic counselling. Since genetic counselling is often not offered by these companies, people seek alternative routes — most commonly the (family) physician, which increases their workload and requires expertise physicians may not have(Mathews et al., 2012). Accordingly, more visits to a physician increase health care costs and become problematic if a significant number of people ask for genetic counselling, as shown by McGuire (2009): 78% of interested consumers would ask their physician for help. Even if it is true that the risk information could be useful, this at least requires us to burden medical resources significantly. Only then are people able to act upon those results.
Assumption 3: You act upon the risk information in a ‘healthy’ way
Risk information is supposed to provide you with the knowledge to act, which increases your opportunities to prevent diseases or, in general, be healthier. When we disregard the point that this information is biased and does not give you the knowledge to act, we can still see two problems with the empowerment claim: people may not act upon receiving risk information, and sometimes there is little that can be done for mitigating these risks..
Do We Change?
It is simply not true that a lot of people act upon risk information. Less than 25% of people engaging in genetic testing change their behaviour, even when studies assume people report accurately and that those changes last long-term (Stewart et al., 2018). If anything, research shows that you become more fatalistic based on an increased risk and blame your bad genes instead of your lifestyle (Marteau & Lerman, 2001).
Less than 25% of people engaging in genetic testing change their behaviour
Additionally, a low risk encourages people to act in unhealthy ways. Note how a low risk is especially harmful because most tests lack validity. I will give two examples of this effect:
- People receiving low-risk test results for alcohol abuse discounted the repercussions of the use of alcohol (Ahn & Perricone, 2022).
- People were less likely to seek medical care for diseases that were classified as low-risk (McCabe & McCabe, 2004).
How Can We Change?
The reason genetic testing is effective for some diseases is that — for those diseases — the impact of lifestyle, environment or other factors is limited anyway. This means that if the genetic test is providing valid information, it is likely there is little that can be done for prevention. This characteristic leaves you with either no opportunity to act at all or stimulated to make rigid interventions.
The reason genetic testing is effective is that the impact of lifestyle, environment or other factors is limited anyway
A good example of the latter is the case of Angelina Jolie, who underwent a mastectomy to decrease her risk of breast cancer (Jolie, 2013). Because examples like Jolie's have led to unnecessarily rigid interventions based on inaccurate information and false positives, the U.S. Preventive Services Task Force classifies testing for breast cancer in the general population as potentially harmful (Moyer, 2014).
Assumption 4: Commercial genetic testing claims advantages over clinical genetic testing
Commercial genetic testing is not unique because genetic testing is already offered within the clinical context. Therefore, it may at this point be ambiguous why we would want commercial testing in the first place. Most commonly, there are two advantages these companies like to claim.
Skipping The Doctor
Companies make access to genetic information more convenient. However, since the information that is accessed is likely not useful, we could ask: ‘Why would this be a good thing?’' Moreover, even if the testing part is more convenient, this is mitigated if you still end up in the medical circuit for counselling or intervention. At best, you save one visit.
Access For Everyone
Companies market to the general public. This gives more people the opportunity to test, which is supposed to lead to fewer costs for society because they discover a high risk earlier (Helgason & Stefánsson, 2010). This contrasts with two points I made before:
- The costs for genetic counselling and additional tests increase the workload for physicians. This comes at the expense of a nation’s social structure.
- The claim that we save costs for society because we discover high risks sooner works the other way as well. People with lower risk may discount their existing risk (e.g. drinking more alcohol because they know to have a low risk for alcohol addiction), making them less likely to be healthier and leading to more costs.
In any case, both advantages of commercial testing are built upon the three preceding assumptions. Once the assumptions don’t hold, neither do the advantages.
An Attempt To End Constructively
Even if we may not like it, the obsession with risk and how to influence it will continue. Therefore, it would be unsatisfying to end on this note. In essence, commercial genetic testing is a product of our need for early and easy access to valid knowledge that can be used to improve our health. Commercial genetic testing in its current state is unable to fulfil that promise.
At least, we need more regulation regarding the marketing, validity and interpretation of commercial genetic testing (Berg & Fryer-Edwards, 2008). Alternatively, we may want to fully integrate genetic testing within the public healthcare system — which requires a significant reduction in costs (Su, 2013).
Even if it is true that the risk information could be useful, this at least requires us to burden medical resources significantly
References
23andMe. (n.d.). 23andMe. Retrieved November 17, 2022.
Ahn, W. kyoung, & Perricone, A. M. (2022). The symptom discounting effect: what to do when negative genetic test results become risk factors for alcohol use disorder. Nature Scientific Reports, 12 (3579), 1–15.
Beck, U. (1992). Risk society : towards a new modernity. Sage Publications.
Berg, C., & Fryer-Edwards, K. (2008). The Ethical Challenges of Direct-to-Consumer Genetic Testing. Journal of Business Ethics, 77 (1), 17–31.
Harvey, A. (2010). Genetic risks and healthy choices: Creating citizen-consumers of genetic services through empowerment and facilitation. Sociology of Health and Illness, 32 (3), 365–381.
Helgason, A., & Stefánsson, K. (2010). The past, present, and future of direct-to-consumer genetic tests. Dialogues in Clinical Neuroscience, 12 (1), 61–68.
Hull, S. C., & Prasad, K. (2001). Reading between the Lines: Direct-to-Consumer Advertising of Genetic Testing. Hastings Center Report, 31 (3), 33–35.
Janssens, A. C. J. W., Gwinn, M., Bradley, L. A., Oostra, B. A., van Duijn, C. M., & Khoury, M. J. (2008). A Critical Appraisal of the Scientific Basis of Commercial Genomic Profiles Used to Assess Health Risks and Personalize Health Interventions. American Journal of Human Genetics, 82 (3), 593–599
Jolie, A. (2013, May 14). My Medical Choice. New York Times.
Lupton, D. (2013). Risk (2nd ed.). Routledge.
Marteau, T. M., & Lerman, C. (2001). Genetic risk and behavioural change. BMJ, 322 (7293), 1056–1060.
Mathews, R., Hall, W., & Carter, A. (2012). Direct-to-consumer genetic testing for addiction susceptibility: A premature commercialisation of doubtful validity and value. Addiction, 107 (12), 2069–2074.
McCabe, L. L., & McCabe, E. R. B. (2004). Direct-to-consumer genetic testing: Access and marketing. Genetics in Medicine, 6 (1), 58–59.
McGuire, A. L., & Burke, W. (2011). Health System Implications of Direct-to-Consumer Personal Genome Testing. Public Health Genomics, 14 (1), 53–58.
McGuire, A. L., Diaz, C. M., Wang, T., & Hilsenbeck, S. G. (2009). Social Networkers’ Attitudes Toward Direct-to-Consumer Personal Genome Testing. American Journal of Bioethics, 9 (6–7), 3–10.
Moyer, V. A. (2014). Risk Assessment, Genetic Counseling, and Genetic Testing for BRCA-Related Cancer in Women: U.S. Preventive Services Task Force Recommendation Statement. Annals of Internal Medicine, 160 (4), 271–282.
Stewart, K. F. J., Wesselius, A., Schreurs, M. A. C., Schols, A. M. W. J., & Zeegers, M. P. (2018). Behavioural changes, sharing behaviour and psychological responses after receiving direct-to-consumer genetic test results: a systematic review and meta-analysis. Journal of Community Genetics, 9, 1–18.
Su, P. (2013). Direct-to-Consumer Genetic testing: A Comprehensive View. Yale Journal of Biology and Medicine, 86, 359–365.
Teladoc Health. (n.d.). Teladoc Health. Retrieved November 17, 2022.
Weedon, M. N., Jackson, L., Harrison, J. W., Ruth, K. S., Tyrrell, J., Hattersley, A. T., & Wright, C. F. (2021). Assessing the analytical validity of SNP-chips for detecting very rare pathogenic variants: implications for direct-to-consumer genetic testing. BMJ, 372.